Physical Properties and Crystal Types
From personal experience you known that solids exhibit a wide
range of physical properties. Some, such as diamond, are very hard;
others, such as naphthalene (moth balls) or ice, are soft, by
comparison, and are easily crushed. Some solids, such as salt crystals
or iron, have high melting points, whereas others, such as candle wax,
melt at low temperatures. Some conduct electricity well, but others are
nonconducting.
Physical properties such as these depend on the kinds and
strengths of the attractive forces that hold the particles together in
the solid. Even though exact predictions cannot be made some
generalizations do exist. In making these generalizations, we can divide
crystals into several types according to the kinds of particles located
at sites in the lattice and the kinds of attractions that exist between
the particles.
Ionic Crystals
Ionic crystals are hard, have high melting points, and are
brittle. When they melt, the resulting liquids conduct electricity well.
These properties reflect the strong attractive forces between ions of
opposite charge as well as the repulsions that occur when ions of like
charge are placed near each other.
They are brittle and tend to shatter into smaller crystals when
stressed. When a crystal is hammered or stressed, ions with like charges
are forced into close proximity. The crystal then literaly self
destructs because of electrostatic repulsion.
Molecular Crystals
Molecular crystals are solids in which the lattice sites are
occupied either by atoms - as in solid argon or krypton - or by
molecules - as in solid CO2, SO2, or H2O.
Such solids tend to be soft and have low melting points because the
particles in the solid experience relatively weak intermolecular
attractions. The crystals are soft because little effort is needed to
separate the particles or to move them past each other. The solid melts
at low temperatures because the particles need little kinetic energy to
break away from the solid. If the crystals contain only individual
atoms, as in solid argon or krypton, or if they are composed of
non-polar molecules, as in naphthalene, the only attractions between the
molecules are the London forces. In crystals containing polar molecules,
such as sulphur dioxide, the major forces that hold the particles
together are dipole-dipole attractions. In crystals such as water the
primary forces of attractions are due to hydrogen bonding.
Covalent Crystals
Covalent crystals are solids in which the lattice points are
occupied by atoms that are covalently bonded to other atoms at
neighbouring lattice sites. The result is a crystal that is essentially
one gigantic molecule. These solids are sometimes called network
solids because of the interlocking network of covalent bonds extending
throughout the crystal in all directions. A typical example is the
diamond, the structure of which is illustrated below.
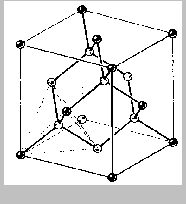
The above structure is diamond. Notice that each carbon atom is
covalently bonded to four others at the corners of a tetrahedron. This
structure extends throughout an entire diamond crystal. (In diamond, of
course, all the atoms are identical. They are different shades here only
to make it easier to visualize the structure.)
Covalent crystals tend to be hard and to have very high melting
points because of the strong attractions between covalently bonded
atoms. They do not conduct electricity because the electrons are bound
too tightly to the bonds. Other examples of covalent crystals are quartz
(SiO2 - typical grains of sand) and silicon carbide (SiC - a
common abrasive used in sandpaper).
Metallic crystals
Metallic crystals have properties that are quite different from
those of the other three types of crystals above. They conduct heat and
electricity well, and they have the lustre that we characteristically
associate with metals. A number of different models have been developed
to describe metals. The simplest one views the crystal as having
positive ions at the lattice positions which are surrounded by electrons
in a cloud that spreads throughout the entire solid.
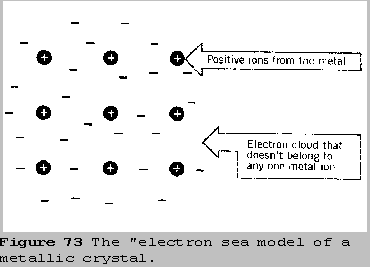
The electrons in this cloud belong to no single positive ion, but
rather to the crystal as a whole. Because the electrons aren't localized
on any one atom, they are free to move easily, which accounts for the
electrical conductivity of metals. By their movement, the electrons can
also transmit kinetic energy rapidly thorough the solid, so metals are
also good conductors of heat. Because the electrons are free to move
easily, even the lattice points are moveable or deformable and this
helps explain the malleability of metals. Even though the lattice points
and electrons are free to move there are some strong attractive forces
at work and these help explain ductility in some metals. This model also
explains the lustre of metals. When light shines on the metal, the
loosely held electrons vibrate easily and readily reemit the light with
essentially the same frequency and intensity.
It is not possible to make simple generalizations about the
melting points of metals. Some have very high melting points, like
tungsten, whereas others like mercury have quite low melting points. To
some degree the melting point depends on the charge of the positive ions
in the metallic crystals. The ions of Group IA tend to exist as cations
with a +1 charge, and they are only weakly attracted to the "electron
sea" that surrounds them. Atoms of Group IIA metals, however, tend to
form cations with a +2 charge. These more highly charged ions are
attached more strongly to the surrounding electron sea, so the Group IIA
metals have higher melting points than their neighbours in Group IA. For
example magnesium melts at 650oC but sodium melts at 98oC.
Tungsten which has a very high melting point, must have very strong
attractions between their atoms, which suggests that there probably is
some covalent bonding in them as well.
Crystal Types
| Type |
Particles Occupying
Lattice Sites
|
Type of
Attractive Force |
Typical
Examples |
Typical
Properties |
| Ionic |
Positive & negative ions |
Attractions between
opposite ions |
NaCl,
NaNO3,
CaCl2 |
Hard; high melting points;
non-conductors of
electricity as solids
but good conductors when
melted
|
| Molecular |
Atoms or Molecules |
Dipole-dipole attractions
London forces
Hydrogen bonding |
HCl
SO2
N2,
Ar ,
CH4
H2O
|
Soft, low melting points;
nonconductors of electricity
in both solid and melted form |
Covalent
(network) |
Atoms |
Covalent bonds
between atoms |
Diamond,
SiC, SiO2 |
Very hard; very high melting pts. nonconductors of
electricity
|
| Metallic |
Atoms |
Positive Attractions
between ions and
an electron cloud
that extends throughout
the crystal in both
solid and liquid |
Cu
Ag
Fe
Na
Hg
Au |
Range from very hard to soft;
melting points range from high
to low; conduct electricity and
heat well; have a characteristic |
|

